(I) KMnO4 + SnCl2 + HCl → MnCl2 + SnCl4 + H2O + KCl
Oxidation half reaction: (loss of electron)
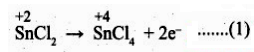
Reduction of halfa reaction: (gain of electron)
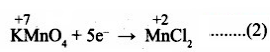
Add H2O to balance oxygen atoms
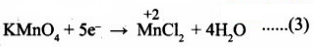
Add Hcl to balance hydrogen atoms
KMnO4 + 5e- + 8 HCl → MnCl2 + 4H2O .......(4)
To equalize the number of electrons equation (1) × 5 and equation (2) × 2
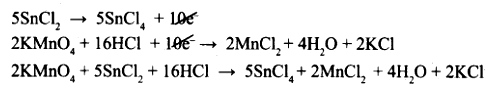
(II) Molecular mass of H3 BO3 = (1 × 3) + (11 × 1) + (16 × 3) = 62
Boric acid sample contains 0.543 mole.
Mass of 0.543 mole of Boric acid = Molecular mass x mole
= 62 × 0.543
= 33.66 g