Correct option (a)
Explanation:
A compound is said to have aromatic character if ring system is planar (with p-orbital) and there is complete delocalisation of π -electrons (lone pair may be taken for delocalisation as relay electrons). This is true only in conjugated cyclic system.
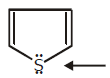
This pair is used in delocalisation
Huckel rule is followed by when electrons used in delocalisation
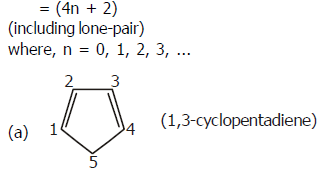
– ring is planar
– ring is not conju
–delocalisation of π -electrons is not possible after C4.
–(4n + 2) π-electrons = 4
Hence, it is not aromatic