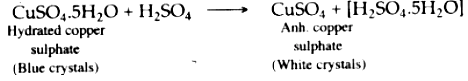
Hydrated copper (II) sulphate, when added to concentrated sulphuric acid, loses water of crystallization and thus white anhydrous copper (II) sulphate is formed. The blue **** hydrated copper (II) sulphate turns white due to the loss of water of crystallization.