In a thermodynamic process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by TΔX, where T is temperature of the system and ΔX is the infinitesimal change in a thermodynamic quantity
X of the system. For a mole of monatomic ideal gas x = - 3/2R in(T/TA) + R in (V/VA). Here, R is gas constant, V is volume of gas, TA and VA are constants.
The List-l below gives some quantities involved in a process and List-II gives some possible values of these quantities.
| List- | List-II |
| (l) Work done by the system in process 1 → 2 → 3 | (P) 1/3RT0 In 2 |
| (II) Change in internal energy in process 1 → 2 → 3 | (Q) 1/3 RT0 |
| (III) Heat absorbed by the system in process 1 → 2→ 3 | (R) RT0 |
| (IV) Heat absorbed by the system in process 1 → 2 | ( *) RT0 |
| | (T) 1/3 RT0(3 + in2) |
| | (U) 5/6RT0 |
If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with P0V0 = 1/3RT0,the correct match is,
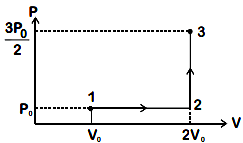
(A) I → Q, II → R, III → *, IV → U
(B) I → Q, II → *, III → R, IV → U
(C) I → *, II → R, III → Q, IV → T
(D) I → Q, II → R, III → P, IV → U