(i) Strength of Acid or Base depends on the extent of its ionisation. Hence equilibrium constant Ka or Kb respectively of the following equilibrium give a quantitative measurement of the strength of the acid or base.
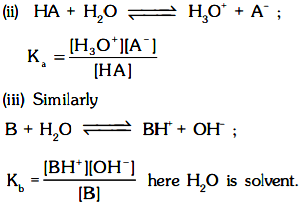
Note :
(i) The other ways to represent above equilibrium is :
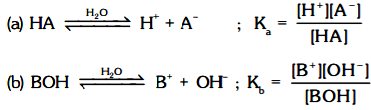
(ii) The larger the value of Ka or Kb, the more complete the ionisation, the higher the concentration of H3O+ or OH– and stronger is the acid or base.