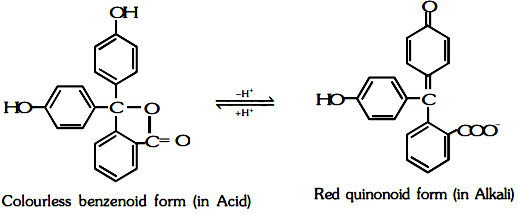
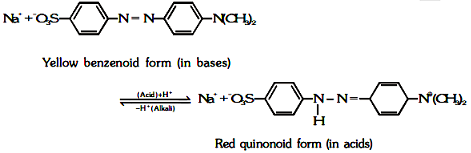
According to this theory,
(i) An acid-base indicator is a dynamic equilibrium mixture of two alternative tautometric forms ; ordinarily one form is benzenoid while the other is quinonoid.
(ii) The two forms have different colours.
(iii) Out of these one form exist in acidic solution, while the other in alkaline solution.
(iv) The change in pH cause the transition of benzenoid form to quinonoid form and vice-versa and consequently change in colour.
Ex: (a) For methyl orange
(b) For Phenolphthalein