(i) Mixture + MnO2 + H2SO4 (conc.)→yellowish greengas
The reaction suggests that the mixture contains Cl- ions.
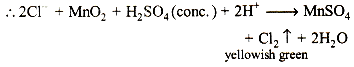
(ii) Mixture + NaOH→Gas turning red litmus blue The reaction suggests that the gas is ammonia (ie, basic in character turning red litmus blue)" Ammonia will be evolved from an ammonium salt. Thus,
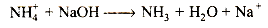
(iii) Solution cf mixture + K3[Fe(CN)6]→blue ppt.
The reaction suggests that the mixture contains a Fe (II) salt. Iron (II) reacts with K3[Fe(CN)6] to give a blue ppt. of Prussian blue complex
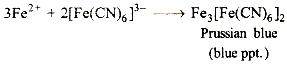
Red colouration with ammonium thiocyanate suggests that some Fe (III) is also present" It is likely that a part of Fe (II) is oxidised to Fe (III) by air
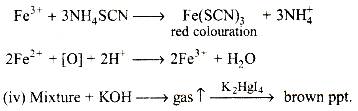
The above reaction can be explained, when ammonia is liberated on boiling the mixture with potassium hydroxide. The liberated ammonia reacts with an alkaline solution of K2HgI4 to give a brown ppt.
Hence, the mixture contains, Fe2+, NH+4, and Cl- ions (with an impurity of Fe3+ ions). The two salts are :
FeCl2 and NH4Cl.